
We've used all the valence electrons, so this is the best structure for ClF3. So in our Lewis structure for ClF3, we have formal charges of zero for each of the atoms. We see that there are six nonbonding around the Fluorines, and then these two right here involved in the chemical bonds. All of the Fluorines are the same, they're symmetrical, so let's just do one of them. It has 4, on the Lewis structure here, nonbonding valence electrons, and then 6 that are involved in chemical bonds, which we divide by 2. 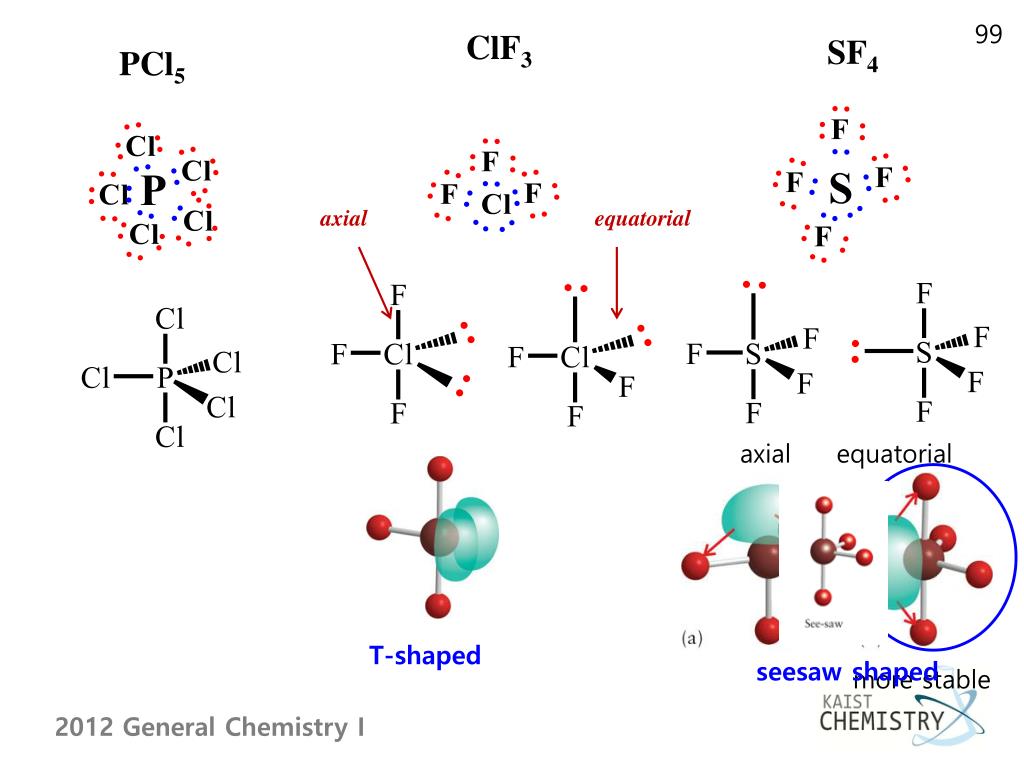 
So we can see on the periodic table, Chlorine has 7 valence electrons. Let's check our formal charges and make sure that this is the best structure for ClF3. We're going to put those two pairs of electrons on the Chlorine, like this. We're going to put those two pairs, those four valence electrons, on the central Carbon-central Chlorine, right here. That means we have 4 valence electrons left over. We've used 6 valence electrons and then around the outside, we've used 6, 8, and 24. Form chemical bonds between the atoms here. Contact with organic materials may result in spontaneous ignition. It reacts with water to form chlorine and hydrofluoric acid with release of heat. We'll put the Chlorine at the center and the Fluorines around the outside. HF Electron Domain Geometry Molecular Geometry Diatomic Diatomic. Chlorine trifluoride appears as a colorless gas or green liquid with a pungent odor. ClF3 has a total of 28 valence electrons. If you examine only the Very Severe Crowdings for a moment, you will see why nature prefers the T-shaped ClF3 structure over the trigonal planar ClF3 structure.Transcript: This is Dr. In Picture T, how many Very Severe Crowdings are there? 4: two for each lone pair in the trigonal plane, because each lone pair in the plane is 90 degrees from one F atom above the plane and one F atom below the plane. In Picture A how many Very Severe Crowdings are there? 6: two for each F atom, because each bond there is 90 degrees from a lone pair above the trigonal plane and 90 degrees from a lone pair below the trigonal plane. A step-by-step explanation of how to draw the ClF3 Lewis Dot Structure (Chlorine trifluoride).For the ClF3 structure use the periodic table to find the total. 
Moreover, let us agree that a lone electron pair takes up more space than a bond, so a 90-degree angle between a lone pair and a bond would represent a Very Severe Crowding. Let me stipulate with you that a bond angle of 90 degrees represents a Crowding, whereas a bond angle of 120 degrees represents only Minor Discomfort. (a) in a trigonal bipyramid, the two axial positions are located directly across from one another, whereas the three equatorial positions are located in a triangular arrangement. Do you want to find out the molecular geometry of ClF3 If yes then check out this video to know the step-by-step method to determine the molecular geometry of any given molecule. Compare this alternative geometry to the T-shape that the book claims, which I will call Picture T. Answer: You are proposing a geometry where three F atoms lie in a trigonal planar arrangement around a central Cl, with one lone pair in an axial position above this plane, and another lone pair in an axial position below this plane.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
